MIT AI System Identifies Protein Fragments to Inhibit Targets
MIT biologists have created a new AI-driven computational method, called FragFold, that can predict which protein fragments will bind to or inhibit specific target proteins. This innovative approach, detailed in a recent publication in the Proceedings of the National Academy of Sciences, could revolutionize how researchers study protein interactions and develop new therapeutic strategies.
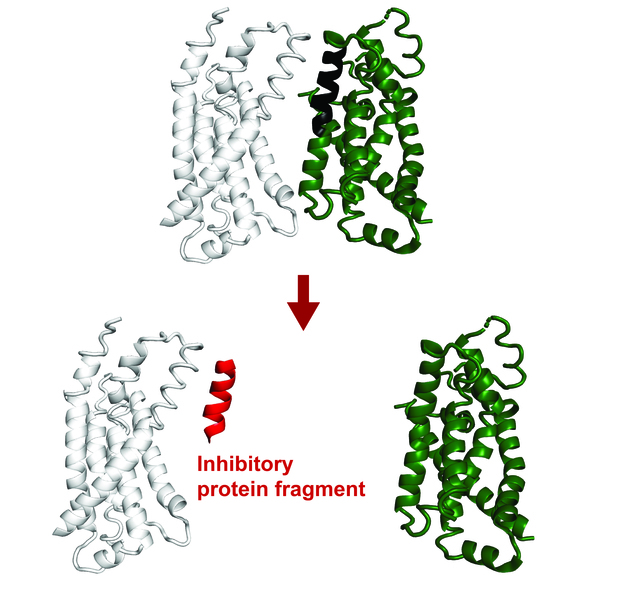
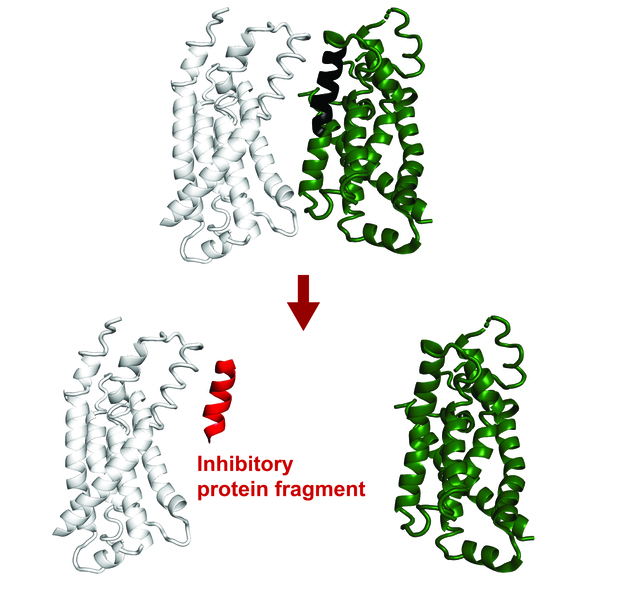
Image of a protein complex. A protein fragment, shown in red, can inhibit the interaction between two other proteins.
Understanding Protein Interactions
All biological functions are determined by how proteins interact with each other. These interactions range from transcribing DNA and controlling cell division, to complex processes in advanced organisms. However, the exact orchestration of these interactions and how proteins interact with other proteins, or even themselves, is still not well understood.
Recent advancements show that small protein fragments possess considerable functional potential. Even though incomplete, short amino acid sequences can bind to the surface of a target protein, replicating natural interactions. This process can change the target protein’s function or disrupt its interactions with other proteins.
Protein fragments could benefit both fundamental research and applied therapeutic research. They can help us understand protein interactions and cellular processes better, and have potential therapeutic applications.
Leveraging Machine Learning
FragFold, developed in the lab of Professor Gene-Wei Li in collaboration with the lab of Professor Amy Keating, utilizes AlphaFold, a machine-learning model that has led to significant breakthroughs in biology. AlphaFold is known for its ability to predict protein folding and interactions. FragFold builds on this and uses it to predict fragment inhibitors, which is a new application.
Researchers verified that more than half of FragFold’s predictions for binding or inhibition were accurate, even when they lacked prior structural data on those interactions.
“Our results suggest that this is a generalizable approach to find binding modes that are likely to inhibit protein function, including for novel protein targets, and you can use these predictions as a starting point for further experiments,” explained Andrew Savinov, co-first and corresponding author and a postdoc in the Li Lab. “We can really apply this to proteins without known functions, without known interactions, without even known structures, and we can put some credence in these models we’re developing.”
To illustrate, one example is FtsZ, a protein key to cell division. This protein has a disordered region, which makes studying it difficult. Disordered proteins are dynamic, and their functional interactions are often fleeting, making them challenging to capture with current structural biology tools. FragFold was used to investigate the activity of FtsZ fragments, revealing several new binding interactions with various proteins. This finding confirmed and extended previous experiments measuring FtsZ’s biological activity.
“This is one example of how AlphaFold is fundamentally changing how we can study molecular and cell biology,” said Keating. “Creative applications of AI methods, such as our work on FragFold, open up unexpected capabilities and new research directions.”
How FragFold Works
The researchers used computation to fragment each protein. They then modeled how these fragments could bind to relevant interaction partners. The maps of these predicted bindings were then compared to the effects of those same fragments in living cells. Millions of cells each produced one type of protein fragment in high-throughput experimental measurements.
AlphaFold uses co-evolutionary information to predict folding using multiple sequence alignments (MSAs) for each prediction. MSAs can be computationally intensive. The researchers in this study pre-calculated the MSA for a full-length protein once, then used that result to guide predictions for each fragment of that protein.
Savinov, along with collaborators including Ph.D. graduate Sebastian Swanson, predicted inhibitory fragments of a range of proteins. This included exploring a complex between lipopolysaccharide transport proteins LptF and LptG. A fragment of LptG inhibited this interaction, seemingly disrupting the delivery of lipopolysaccharide, which is essential for the outer cell membrane of E. coli.
Savinov stated, “The big surprise was that we can predict binding with such high accuracy and, in fact, often predict binding that corresponds to inhibition. For every protein we’ve looked at, we’ve been able to find inhibitors.”
Future Applications
The team initially focused on protein fragments as inhibitors because it was a relatively simple outcome to measure in a system. Moving forward, Savinov is exploring fragment function beyond inhibition. Fragments could stabilize the protein they bind to, enhance or alter function, or even trigger protein degradation.
This research offers a starting point for developing a systemic understanding of cellular design principles and what elements deep-learning models may be using to make accurate predictions.
“There’s a broader, further-reaching goal that we’re building towards,” Savinov said. “Now that we can predict them, can we use the data we have from predictions and experiments to pull out the salient features to figure out what AlphaFold has actually learned about what makes a good inhibitor?”
The researchers also analyzed in depth how protein fragments bind, exploring different protein interactions and mutating specific residues to see how they interact with their target. They examined the behavior of many mutated fragments within cells, called deep mutational scanning, revealing some key amino acids involved in inhibition.
“The core strength of this work is the interplay between high-throughput experimental inhibition data and the predicted structural models: the experimental data guides us towards the fragments that are particularly interesting, while the structural models predicted by FragFold provide a specific, testable hypothesis for how the fragments function on a molecular level,” according to Swanson.
Li added, “By creating compact, genetically encodable binders, FragFold opens a wide range of possibilities to manipulate protein function…We can imagine delivering functionalized fragments that can modify native proteins, change their subcellular localization, and even reprogram them to create new tools for studying cell biology and treating diseases.”